Jean-Pierre Changeux is France’s most famous neuroscientist. Though less well known in the United States, he has directed a famous laboratory at the Pasteur Institute for more than thirty years, taught as a professor at the Collège de France, and written a number of works exploring “the neurobiology of meaning.” Aside from his own books, Changeux has published two wide-ranging dialogues about mind and matter, one with the mathematician Alain Connes and the other with the late French philosopher Paul Ricoeur.
Changeux came of age at a fortunate time. Born in 1936, he began his studies when the advent both of the DNA age and of high-resolution images of the brain heralded a series of impressive breakthroughs. Changeux took part in one such advance in 1965 when, together with Jacques Monod and Jeffries Wyman, he established an important model of protein interactions in bacteria, which, when applied to the brain, became crucial for understanding the behavior of neurons. Since that time, Changeux has written a number of books exploring the functions of the brain.
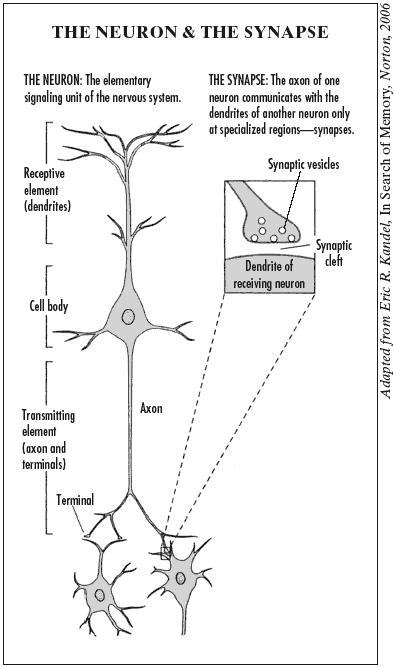
The brain is of course tremendously complex: a bundle of some hundred billion neurons, or nerve cells, each sharing as many as ten thousand connections with other neurons. But at its most fundamental level, the neuron, the brain’s structure is not difficult to grasp. A large crown of little branches, known as “dendrites,” extends above the body of the cell and receives signals from other neurons, while a long trunk or “axon,” which conducts neural messages, projects below, occasionally shooting off to connect with other neurons. The structure of the neuron naturally lends itself to comparison with the branches, trunk, and roots of a tree, and indeed the technical term for the growth of dendrites is “arborization.” (See the illustration below.)
We’ve known since the early nineteenth century that neurons use electricity to send signals through the body. But a remarkable experiment by Hermann von Hermholtz in 1859 showed that the nervous system, rather than telegraphing messages between muscles and brain, functions far slower than copper wires. As Changeux writes,
Everyday experience leads us to suppose that thoughts pass through the mind with a rapidity that defies the laws of physics. It comes as a stunning surprise to discover that almost the exact opposite is true: the brain is slow—very slow— by comparison with the fundamental forces of the physical world.
Further research by the great Spanish anatomist Santiago Ramon y Cajal suggested why the telegraph analogy failed to hold: most neurons, instead of tying their ends together like spliced wires, leave a gap between the terminus of the neuron, which transmits signals, and the receptor of those signals in the adjacent neuron. How signals from neurons manage to cross this gap, later renamed the synaptic cleft (“synapse” deriving from the Greek for “to bind together”), became the major neurophysiological question of the early twentieth century.
Most leading biologists at that time assumed that neurons would use the electricity in the nervous system to send signals across the cleft. The average synaptic cleft is extremely small—a mere twenty nanometers wide—and though the nervous system may not function at telegraphic speed, it was not difficult to imagine electrical pulses jumping the distance. Further, given the speed with which nerves react, the alternative theory, that electrical pulses would cause a chemical signal to move across the cleft, seemed to rely on far too slow a mechanism. But as the decades passed, hard evidence slowly accumulated in support of the chemical theory. According to Changeux, experiments began to suggest that “the human brain therefore does not make optimal use of the resources of the physical world; it makes do instead with components inherited from simpler organisms…that have survived over the course of biological evolution.”
A remarkable experiment by Otto Loewi in the 1920s first suggested how the brain makes use of its evolutionary inheritance in order to communicate. Loewi bathed a frog’s heart in saline solution and stimulated the nerve that normally slows the heartbeat. If the slowing of the heart was caused by a chemical agent rather than an electrical impulse, Loewi reasoned, then the transmitting chemical would disperse throughout the solution. Loewi tested his hypothesis by placing a second heart in the solution. If nerve transmission was chemical rather than electrical, he supposed, then the chemical slowing down the first heart, dispersed throughout the solution, would likewise slow down the second heart. This is exactly what happened. Loewi named the substance released by the relevant nerve, called the vagus nerve, Vagusstoff; today it is known as the neurotransmitter acetylcholine. By the 1950s, further experiments had definitively proved that most neurons, while using electricity internally, must resort to chemicals to cross the synaptic cleft and communicate with the next neuron in the chain.
Advertisement
Changeux began his work at this stage, when the basic methods for neuron communication had been determined but the detailed chemical mechanisms were just opening up to research. Thanks to new high-resolution images from electron microscopes, first taken by Sanford Palay and George Palade in 1955, biologists could finally see the minute structures of the synapse. They discovered that the transmitting end of the neuron, called the nerve terminal, comes packed with tiny sacs, or vesicles, each containing around five thousand molecules of a specialized chemical, the neurotransmitter. When an electrical signal moves down the neuron, it triggers the vesicles and floods the synaptic cleft with neurotransmitter molecules. These chemical neurotransmitters then attach to the proteins called receptors on the surface of the neuron that is located just across the synaptic cleft, opening a pore and allowing the electrically charged atoms called ions to flow into the neuron. Thus, the chemical signal is converted back into an electrical signal, and the message is passed down the line.
These processes were still somewhat mysterious in 1965, when the young Changeux, working with his teacher Jacques Monod and the American scientist Jeffries Wyman, produced one of the theories for which he became best known. The three scientists, then studying metabolism, attempted to explain how the structure of an enzyme could stabilize when another molecule attached to it. Changeux later saw a parallel with the nervous system. When a chemical neurotransmitter binds to a receptor it holds the ion pore open, ensuring its continuing function, a critical step in converting the neurotransmitter’s chemical signal back into an electrical pulse. Changeux’s discovery established the groundwork for the way many neurons communicate, and his findings were based on the more general paper he had coauthored with Wyman and Monod.
With a working theory for neuron communication established, Changeux then turned to the ways that larger structures in the brain might change these basic interactions. A longstanding theory, introduced by Donald Hebb in 1949, proposed that neurons could increase the strength of their connection through repeated signals. According to a slogan describing the theory, “neurons that fire together, wire together.” Repeated neuron firings, Hebb believed, would produce stronger memories, or faster thought patterns. But researchers found that certain regulatory networks could achieve far more widespread effects by distributing specialized neurotransmitters, such as dopamine and acetylcholine, throughout entire sections of the brain, reinforcing connections without the repeated firings required by Hebb.
Changeux focused on these specialized distribution networks. It was long known that nicotine acts on the same receptor as the neurotransmitter acetylcholine. Changeux recognized that this could explain both nicotine’s obvious benefits—greater concentration, relaxation, etc.—as well as the drug’s more puzzling long-term effects. For instance, while cigarettes are dangerous to health, some studies show that smokers tend to suffer at significantly lower rates from Alzheimer’s disease and Parkinson’s disease. Changeux found that nicotine, by attaching to the same receptors as acetylcholine, reproduces some of the benefits of acetylcholine by reinforcing neuronal connections throughout the brain. Nicotine is not exactly the same chemically as acetylcholine, but can mimic its effects. Changeux’s lab has since focused on the workings of the nicotine/acetylcholine system, and he has attempted to explain how all such regulatory systems, working together, can produce the experience we call consciousness—as well as more abstract concepts like truth.
How, then, does the mass of cells in the brain produce our experience of sight, sound, and imagination? According to Changeux, the infant brain is not a blank slate, receiving all experience and instruction—both what it sees and how to think about it—from the outside. Nor is the infant brain preprogrammed, its reactions predetermined, unable to change itself and adapt. Rather, as Changeux began to hypothesize in the late 1970s, the brain, beginning in the embryo, produces, by means of genetic action, “mental objects of a particular type that might be called prerepresentations—preliminary sketches, schemas, models.”
According to this theory, spontaneous electronic activity in the brain, “acting as a Darwinian-style generator of neuronal diversity,” creates dynamic, highly variable networks of nerve cells, whose variation is comparable with the variation in DNA. Those networks then give rise to the reflex movements of the newborn infant. Over time the infant’s movements become better coordinated. Neural networks associated with more successful movements—such as grasping an object—are “selected”; that is, their activity is reinforced as their synaptic junctions become strengthened. As the child continues to explore his or her surroundings, Darwinian competition strengthens some of these transient networks sufficiently to make them relatively permanent parts of the child’s behavioral repertoire. Changeux calls the process, first elaborated in a 1976 paper, “learning by selection.”
Animals and infants conduct this miniature version of natural selection by means of what Changeux terms “cognitive games.” One well-known example concerns cries of alarm in African vervet monkeys. Adult monkeys use a simple but effective vocabulary of sounds that warn against danger: a loud bark for leopards, a two-syllable cough for eagles, and a hissing sound for snakes. Surprisingly, researchers found, baby monkeys hiss at snakes without explicit instruction. Changeux writes, “Snakes seem to arouse a sort of innate universal fear, which probably developed fairly early in the course of the evolution of the higher vertebrates.” When adult monkeys confirm the baby’s judgment with their own hisses, the infant’s genetically produced prerepresentation is rewarded and reinforced.
Advertisement
But baby monkeys require more explicit instruction in protecting themselves against predators, such as eagles, to which they have been less genetically conditioned. At first,
newborn monkeys react to any form that flies in the air, which is to say to the class of birds as a whole. Then, gradually, a selective stabilization of the response to the shape of dangerous species takes place…. If the first cry of alarm is sounded by one of the young, the nearest adult looks up. If it sees a harmless bird, it does not react. But if the young monkey has spotted a martial eagle, the adult reacts by emitting a cry of alarm that confirms the presence of danger…. The adult’s cry of alarm validates a pertinent relationship between shape and sound that is established in the brain of the young monkey.
This process of learning alarm cries through trial and error, reward and suppression, demonstrates the kind of cognitive games that are played out constantly through the brain’s interaction with the environment. As successful behaviors increase in number, Changeux believes, they strengthen the capacity to consciously manipulate the environment. Most actions are not beneficial, and as each neuron competes for limited resources, many of the least useful neurons literally die out. Changeux therefore hypothesizes: “To learn is to eliminate.”
In Changeux’s view, starting in the womb, spontaneous electrical activity within neurons creates highly variable networks of nerve cells; the networks are selected and reinforced by environmental stimuli; and these reinforced networks can then be said to “represent” the stimuli—e.g., the appearance of a predator—though no particular network of nerve cells exclusively represents any one set of stimuli. The environment does not directly “instruct” the brain; it does not imprint precise images in memory. Rather, working through our senses, the environment selects certain networks and reinforces the connections between them.1
Critical to this process of selection, in Changeux’s view, is the brain’s reward system: the pleasure response. Dopamine is part of a reward system that is important in human and animal behavior, and dopamine levels are elevated in the brain when we experience pleasure or well-being. Pleasure is associated both with the anticipation of activities essential to survival—for example, eating and sex—and with the activities themselves. Changeux describes one particularly telling experiment:
When a trained monkey succeeds in grasping a peanut hidden in a box, the inside of which it cannot see, the activity of dopamine neurons increases at precisely the moment when the animal recognizes the food with its fingers.
But opiates, alcohol, cannabinoids, nicotine, and other drugs can also increase the release of dopamine and subvert the normal function of the reward system. A rat given infusions of cocaine into the brain following the pressing of a bar will persist in pressing the bar repeatedly in preference to consuming food or water. Sugar, too, can be addictive. Indeed, the National Institutes of Health is now studying whether foods high in fat and sugar should be classified as addictive agents, in the same category as nicotine, alcohol, and cocaine.
In general, behaviors associated with pleasure are reinforced by the release of dopamine; as a result, the synaptic junctions of the associated neuronal networks are strengthened. And as they are strengthened, the changes in brain function often become permanent. A former cocaine addict who has been able to live without the drug for a decade may experience an irresistible need for cocaine when returning to a place whose cues evoke past drug-taking experiences. But the memories that are evoked are reconstructions:
Every evocation of memories is a reconstruction on the basis of physical traces stored in the brain in latent form, for example at the level of neurotransmitter receptors.
Instead of recalling the experiences of both pleasure-filled high and painful withdrawal, the addict’s memories may be overwhelmed by the powerful neural connections previously created by the drug. Only if memory is a matter of reconstruction of latent physical traces, not direct recall of past events, Changeux argues, could these kind of drug-induced long-term compulsions occur.
In his book The Physiology of Truth, Changeux connects memory to the acquisition of knowledge and the testing of its validity, as is done in science in general. “We now find ourselves in a position,” Changeux writes,
to sketch the outlines of a plausible interpretation of the neural bases of meaning. The naive view that the neural representation of a complex meaning—a yellow Renault, for example—is located in a single, hierarchically prominent nerve cell…has been found to be unjustified for the most part. It is generally accepted today that distinct populations of neurons in sensory, motor, associative, and other territories are linked as part of a distributed network…[which] mobilizes several distinct and functionally specific territories in a discrete manner, thus constituting a neural embodiment of meaning. Note that this assumption does not require that…anatomical connections be…reproducible across individual brains in every detail [in order to evoke memory], only that a map of functional relations be established.
In some ways Changeux’s ideas are similar to Gerald Edelman’s theory of neural Darwinism. For both Changeux and Edelman, Darwinian selection is an essential part of how the brain functions. And yet Edelman and Changeux have radically different views of what selective mechanisms in the brain imply about the nature of brain function, knowledge, memory, and consciousness. Our senses, in Edelman’s view, are confronted by a chaotic, constantly changing world that has no labels. The brain must create meaning from that chaos. Edelman writes, in A Universe of Consciousness, his book with Giulio Tononi:
It is commonly assumed that memory involves the inscription and storage of information, but what is stored? Is it a coded message? When it is “read out” or recovered, is it unchanged? These questions point to the widespread assumption that what is stored is some kind of representation. [We take] the opposite viewpoint, consistent with a selectionist approach, that memory is nonrepresentational.2
While Changeux also considers selection to be essential to the formation of memory, he, as opposed to Edelman, believes that once a set of neuronal circuits have been selected to form a memory, they become part of a relatively stable structure that “can be conceived as a set of long-lasting global representations.” Though “the precise patterns of connectivity in the network may vary from individual to individual,” Changeux writes,
its functional relationships (or stabilized meanings) remain constant. In this way a “scale model” of external reality…is selected and stored in memory in the brain. Memory objects enjoy a genuine existence, then, as latent “forms” composed of stable neuronal traces.
Changeux says memories can be modified by the addition of new information, or “by preexisting knowledge or by the emotional resonance of actual memories of past experience.”
In contrast to Changeux’s account, Edelman, we believe, has a different and considerably deeper view of memory and what it tells us about the nature of meaning and brain function. Both Changeux and Edelman propose that during memory formation, our interactions with the world cause a Darwinian selection of neural circuits, much as the body, when invaded by a virus, “selects” the most potent antibodies from the enormous repertoire of antibodies made available by the body’s immune system. However, the resulting memory is not, Edelman says, a representation of the outside world, any more than the antibody that has protected the body against an infecting virus is a representation of that virus. Yet the antibody can protect the body against a future attack by the virus, just as the neural circuits can contribute to memory recall. Instead, Edelman writes, memory is the ability to
repeat a mental or physical act after some time despite a changing context…. We stress repetition after some time in this definition because it is the ability to re-create an act separated by a certain duration from the original signal set that is characteristic of memory. And in mentioning a changing context, we pay heed to a key property of memory in the brain: that it is, in some sense, a form of constructive recategorization during ongoing experience, rather than a precise replication of a previous sequence of events.
For Edelman, then, memory is not a “small scale model of external reality,” but a dynamic process that enables us to repeat a mental or physical act:
the key conclusion is that whatever its form, memory itself is a [property of a system]. It cannot be equated exclusively with circuitry, with synaptic changes, with biochemistry, with value constraints, or with behavioral dynamics. Instead, it is the dynamic result of the interactions of all these factors acting together, serving to select an output that repeats a performance or an act.
The overall characteristics of a particular performance may be similar to previous performance, but the ensembles of neurons underlying any two similar performances at different times can be and usually are different. This property ensures that one can repeat the same act, despite remarkable changes in background and context, with ongoing experience.
The validity of the respective approaches of Changeux and Edelman remains to be tested by further inquiry into brain function. The detailed neurophysiological processes involved are still largely unexplored.
In fact, “external reality” is a construction of the brain. Our senses are confronted by a chaotic, constantly changing world that has no labels, and the brain must make sense of that chaos. It is the brain’s correlations of sensory information that create the knowledge we have about our surroundings, such as the sounds of words and music, the images we see in paintings and photographs, the colors we perceive: “perception is not merely a reflection of immediate input,” Edelman and Tononi write, “but involves a construction or a comparison by the brain.”
For example, contrary to our visual experience, there are no colors in the world, only electromagnetic waves of many frequencies. The brain compares the amount of light reflected in the long (red), middle (green), and short (blue) wavelengths, and from these comparisons creates the colors we see. The amount of light reflected by a particular surface—a table, for example— depends on the frequency and the intensity of the light hitting the surface; some surfaces reflect more short-wave frequencies, others more long-wave frequencies. If we could not compare the presence of these wavelengths and were aware of only the individual frequencies of light—each of which would be seen as gray, the darkness or lightness of each frequency depending on the intensity of the light hitting the surface—then the normally changing frequencies and intensities of daylight (as during sunrise, or when a cloud momentarily blocks out the sun) would create a confusing picture of changing grays. Our visual worlds are stabilized because the brain, through color perception, simplifies the environment by comparing the amounts of lightness and darkness in the different frequencies from moment to moment.3
The problem of representation, meaning, and memory is also illustrated by the case of a patient who has lost his arm in an accident. As is often the case, the brain creates a “phantom” limb in an apparent attempt to preserve a unified sense of self. For the patient, the phantom limb is painful. The brain knows there is no limb; pain is the consequence of the incoherence between what the brain “sees” (no arm) and the brain’s “feeling” the presence of a phantom that it has created in its attempt to maintain a unified sense of self in continuity with the past. Such pain is not created by an external stimulus and cannot be eliminated by painkillers.
One famous case is that of a young man who had lost his hand in a motorcycle accident. In a therapeutic procedure devised by V.S. Ramachandran, and described in his book with Sandra Blakeslee, Phantoms in the Brain, the patient put his intact hand in one side of a box and “inserted” his phantom hand in the other side. As the illustration on this page shows, one section of the box had a vertical mirror, which showed a reflection of his intact hand. The patient observed in the mirror the image of his real hand, and was then asked to make similar movements with both “hands,” which suggested to the brain real movement from the lost hand. Suddenly the pain disappeared. Though the young man was perfectly aware of the trick being played on him —the stump of his amputated arm was lying in one section of the box—the visual image overcame his sense of being tricked. Seeing is believing! Pain—the consequence of the incoherence between the brain’s creation of a phantom limb and the visual realization that the limb does not exist—disappeared; what was seen (a hand in the mirror) matched what was felt (a phantom).

According to the Italian neurologist Angela Sirigu, who used videos instead of mirrors to perform a similar experiment,
It is the dissonance between the image of oneself and the damaged body, that is at the origin of the phantom pain. Seeing the damaged hand once again functioning, reduces the dissonance even though the patient is aware of being tricked.
At one moment the patient experiences a painful phantom limb; at another he sees a mirror image of his intact hand and the pain disappears. This is only one of many neurological examples of what we might call the Dr. Jekyll and Mr. Hyde Syndrome: the patient in the experiment sees and remembers one world at certain times and a completely different world at other times.4 The phantom limb is the brain’s way of preserving a body image—a sense of self that is essential to all coherent brain activity. And as in the case of colors, the phantom limb suggests that what we see, hear, and feel are inventions of the brain—an integration of the past (the loss of the limb) and the present (a phantom that is essential for the brain’s continuing to function “normally”).
In general, every recollection refers not only to the remembered event or person or object but to the person who is remembering. The very essence of memory is subjective, not mechanical, reproduction; and essential to that subjective psychology is that every remembered image of a person, place, idea, or object inevitably contains, whether explicitly or implicitly, a basic reference to the person who is remembering.
Our conscious life is a constant flow, or integration, of an immediate past and the present—what Henri Bergson called le souvenir du présent (1908) and Edelman more recently called the remembered present (1989). Consciousness, in this view, is neither recalled representations nor the immediate present, but something different in kind (as colors are different in kind from the lightness and darkness of different reflected wavelengths).
The importance of body image and motor activity for perception, physical movement, and thought is suggested by the recent discovery of “mirror neurons” by Giacomo Rizzolatti and his colleagues. They observed that the neurons that fired when a monkey grasped an object also fired when the monkey watched a scientist grasp the same object. The monkey apparently understood the action of the experimenter because the activity within its brain was similar when the monkey was observing the experimenter and when the monkey was grasping the object. What was surprising was that the same neurons that produced “motor actions,” i.e., actions involving muscular movement, were active when the monkey was perceiving those actions performed by others.
The “rigid divide,” Rizzolatti and Corrado Sinigaglia write in their new book, Mirrors in the Brain,
between perceptive, motor, and cognitive processes is to a great extent artificial; not only does perception appear to be embedded in the dynamics of action, becoming much more composite than used to be thought in the past, but the acting brain is also and above all a brain that understands.
We can recognize and understand the actions of others because of the mirror neurons; as Rizzolatti and Sinigaglia write, this understanding “depends first of all on our motor neurons.”5 Our abilities to understand and react to the emotions of others may depend on the brain’s ability to imitate the neuronal activity of the individual being observed.
When we see a friend crying, we may feel sympathy because the activity in our brain is similar to that in the brain of the person crying. We recognize disgust in another person through our own experience of the feeling of disgust and the associated neural activity.
Rizzolatti and Sinigaglia write:
our perceptions of the motor acts and emotive reactions of others appear to be united by a mirror mechanism that permits our brain to immediately understand what we are seeing, feeling, or imagining others to be doing, as it triggers the same neural structures… that are responsible for our own actions and emotions.
The nature of the brain’s “representations”—if there is such a thing—of the world, the self, the past and present, remains puzzling, as the very different approaches we have described suggest: Changeux’s view of “long-lasting global representations”; Edelman and Tononi’s view of memory as constructive recategorizations, and Rizzolatti’s stunning discovery of mirror neurons, suggesting that we know and understand others, to some extent, through neural imitation.
And as these differing views show, while we are still far from a full understanding of the nature of memory, perception, and meaning, it is nonetheless because of the work of scientists such as Changeux, Edelman, and Rizzolatti that we have a better grasp of the complexity of subjective experiences. Perhaps in the future, questions about higher brain functions will be better understood because of new genetic and neurophysiological discoveries and brain imaging. An unexpected scientific discovery can give us a new insight into something we thought we had always known: mirror neurons, Rizzolatti tells us, “show how strong and deeply rooted is the bond that ties us to others, or in other words, how bizarre it would be to conceive of an I without an us.”
This Issue
June 26, 2008
-
1
Neural circuits selected during memory formation may be strengthened by the addition of new neurotransmitter receptors to the synaptic junctions. This is called Long Term Potentiation (LTP). The weakening or elimination of circuits can lead to memory loss, which occurs normally with aging but is accelerated in neurodegenerative diseases such as Alzheimer’s. In that disease, neurons in the hippocampus, a brain region that is important to memory function, as well as other neurons in the brain, lose their synapses and eventually die, leading to memory impairment. Despite extensive investigation, the cause of neuron death in Alzheimer’s disease is not understood. (Some of the recent research on memory loss is mentioned by Sue Halpern in “Memory: Forgetting Is the New Normal,” Time, May 8, 2008.) ↩
-
2
For further discussion see Israel Rosenfield, “Neural Darwinism: A New Approach to Memory and Perception,” The New York Review, October 9, 1986, as well as The Invention of Memory (Basic Books, 1988). ↩
-
3
See Oliver Sacks and Robert Wasserman, “The Case of the Colorblind Painter,” The New York Review, November 19, 1987. ↩
-
4
See Israel Rosenfield, L’étrange, le familier, l’oublié (Paris: Flammarion, 2005) for further discussion; and Reilly et al., “Persistent Hand Motor Commands in the Amputees’ Brain,” Brain (August 2006), for evidence that the brain is maintaining normal motor commands despite the loss of a limb. ↩
-
5
V.S. Ramachandran believes that mirror neurons might give us further clues to the nature of phantom limb pain. He has noted that phantom pain disappeared for ten or fifteen minutes when a patient was observing a volunteer rub her hand and he has suggested that the suppression of pain in such cases might involve the mirror neurons. However, the mechanism of the pain suppression is not clear. ↩


