Let me say before I go any further that I forgive nobody.
—Samuel Beckett
Quantum mechanics is the most accurate and most general scheme for making predictions about the behavior of physical systems in the history of natural science. Its arrival in the 1920s reduced the entire field of chemistry, more or less overnight, to a special application of the laws of physics, and suddenly made it possible to understand the shining of stars, the conduction of electricity, and the very existence and stability of familiar material things. It has been indispensable to the development of many of the twentieth-century technologies that have so radically transformed modern life and, most importantly for our purposes here, it is the logical foundation upon which the entirety of physics is carried out.
But controversies about what quantum mechanics implies, about both the nature of the world and the nature of science, have been going on for almost a hundred years, and have been the focus of enormous interest not only in theoretical physics but in philosophy, popular media, and other numerous and far-flung corners of contemporary culture as well. The history of these controversies is the subject of Adam Becker’s new book, What Is Real?
The story that Becker tells (and this is perhaps the place to say that he interviewed me in the course of his research, and that I am both mentioned and thanked in his book) runs, in a nutshell, like this: Once upon a time, physics aspired to offer us an objective, literal, realistic, comprehensive, and mechanical account of what the world is actually like. But that aspiration suddenly began to look quaint, naive, and out of date in the early decades of the twentieth century, under the assault of new and unsettling discoveries about the behaviors of subatomic particles.
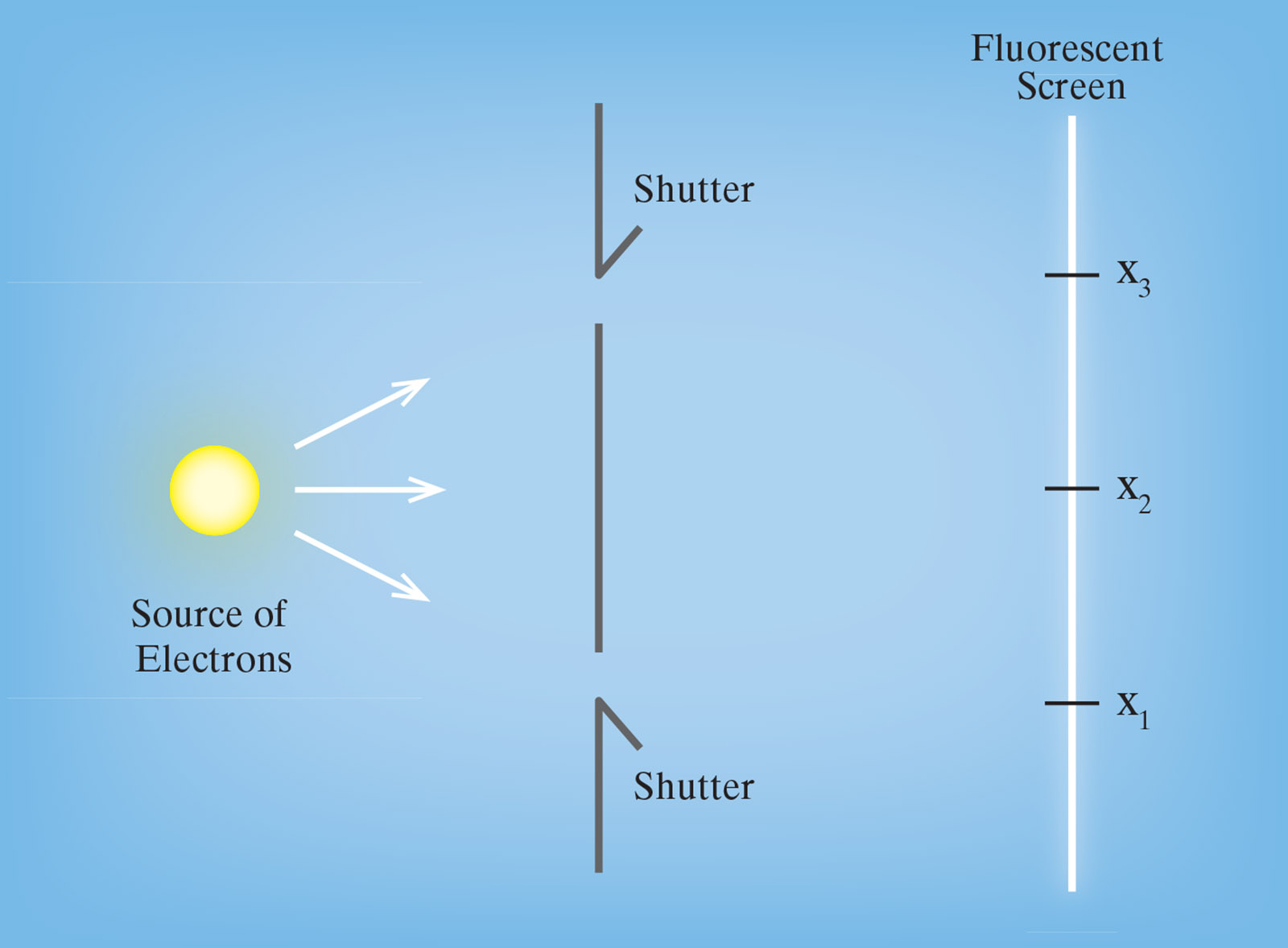
One famous example is the “double-slit” experiment: on the left (as shown in the figure below) there is a source of electrons, and in the middle there is a screen through which electrons cannot pass. The screen has two slits in it, both of which can be covered with shutters. On the right is a second screen, which is coated with a fluorescent material that lights up for a moment at the point where an electron strikes it.
Let’s consider three different cases. (1) Suppose that we cover one of the slits and allow a fairly large number of electrons to stream out of the source. Some of those electrons will make it to the fluorescent screen and reveal themselves there as little flashes of light—and if you make a graph of the number that land in each particular location on the fluorescent screen, it will form a bell-shaped curve centered on x1, if you have covered the top slit, or on x3, if you have covered the bottom one. (2) If we send some electrons through with only the top slit open and then send some others through with only the bottom slit open, and look at the pattern in which the electrons accumulate on the screen, what we will find is a pair of bell-shaped curves—one centered on x1 and the other centered on x3. (3) If we send a large number of electrons through with both slits simultaneously open, we naturally expect that roughly half of the ones that get to the fluorescent screen go through the upper slit and roughly half go through the lower slit, so that the overall pattern is again going to be a pair of bell-shaped curves, as it was in case (2).
But that, astonishingly, is not what happens. When both slits are simultaneously open, we still get large bumps at x1 and x3, but the pattern of landings now also contains a collection of smaller bumps and dips in the vicinity of x2—something physicists call an interference pattern—which simply aren’t there in the pair of bell-shaped curves that we got in case (2). We might think that the difference between cases (2) and (3) has something to do with collisions between electrons that pass through the top slit and electrons that pass through the bottom one. This, however, isn’t the case: it can easily be confirmed that we get the same interference pattern, and not the less complicated pair of bell-shaped curves that we got in case (2), even if we slow down the emissions of electrons so that no more than one electron is ever anywhere in the device at any given time.
So it seems as if an electron that makes it from the source to the fluorescent screen when both slits are simultaneously open must not be passing through one of those slits or the other but rather, somehow, through both. And yet, if we attach electron-detectors to the slits, and if we send one electron through the device, then we either detect it passing through the lower slit and find nothing at the upper one, or we detect it passing through the upper slit and find nothing at the lower one. On top of that it turns out that the business of merely looking, of merely producing a record of which of the two slits each particular electron goes through, somehow causes the pattern of landings to revert to the more sensible pair of bell-shaped curves from case (2).
Advertisement
Notwithstanding the obvious strangeness of all this, a concise and efficient set of rules was discovered that turned out to be extraordinarily successful at predicting the behaviors of electrons in circumstances like these, and that has since turned out to be extraordinarily successful at predicting the behaviors of all of the physical systems with which we are familiar, under all of the physical conditions that we have encountered. And this peculiar phenomenon of “interference” turns out to be no mere curiosity. It is absolutely essential to the microscopic functioning of nature. For example, if not for precisely the sorts of interference effects that make the double-slit experiment so puzzling, lasers would not function, metals would not conduct electricity, there would be no such thing as chemical combination, and atoms themselves would quickly cease to exist. The rules that predict all that are called quantum mechanics.
But the business of descrying anything like a picture of the world in this set of rules seemed very daunting. Think, for example, of the electrons in case (3). How exactly do they get from the source to the fluorescent screen? It doesn’t seem right to think of each electron as passing through either the upper slit or the lower one, because we know (from the experiments in which we only leave one slit open at a time, and also from the experiments in which the detectors are switched on) that a set of electrons, each of which passes either through the upper slit or through the lower one, is going to land on the fluorescent screen in the double-bell-curve pattern of case (2), which is not the pattern we get in case (3). But it also doesn’t seem right to think of each electron as somehow passing through both the upper slit and the lower one—because we know that if we were to turn the detectors on we would surely find an electron at exactly one of the two slits and nothing whatever at the other. And they certainly cannot have gotten from the source to the fluorescent screen by any other route.
There was a brilliant circle of physicists around Niels Bohr, at his institute in Copenhagen, who had been at the forefront of the mathematical development of quantum mechanics from its beginnings, and who had managed to persuade themselves, by sometime around the mid-1920s, that all of this was going to require, as Bohr put it, some “radical revision of our attitude toward the problem of physical reality.” They were convinced that any attempt at describing what is “actually going on” in the double-slit experiment, or describing what things are “actually like” in the interiors of atoms, must inevitably collapse into paradox. What was happening, they thought, was nothing less than the scientific discovery of the limits of science itself. Physics, as they saw it, could no longer pretend to be in the business of finding out how nature is. As Becker writes, for Bohr “quantum physics tells us nothing whatsoever about the world…because quantum objects don’t exist in the same way as the everyday world around us.” What physics is supposed to do, and all that physics can aspire to do—according to Bohr and his circle—is make predictions about the results of experiments.
Spelling out the details of this new conception of physics—the so-called Copenhagen interpretation—turns out to be tricky. And the various attempts we have at doing so, from Bohr and his circle, are not always obviously compatible with one another, or even internally consistent in themselves. One of the things that seems clear is that they were thinking of quantum mechanics as a universal theory, which could in principle be applied to any physical system. But that came with a curious catch, which was that any application of the mathematical apparatus of quantum mechanics to some particular physical system X is apparently going to need to allude to something else, something outside of X, by which X can be measured or observed. And this external observer needs to be described, according to Bohr and his circle, for reasons that were never fully explained, in the more familiar and everyday language of “classical” physics.1
Any application of quantum mechanics, then, is going to need to start by drawing a line between the system that gets measured (to which one applies the mathematical formalism of quantum mechanics) and the system that does the measuring (which needs, again, to be treated as “classical”). And there is no objective fact of the matter, out there in the world, about where that line belongs. Where it gets drawn is going to depend on what you have chosen to treat as the system that gets measured and what you have chosen to treat as the system that does the measuring. You can draw the line between some subatomic object of experimental investigation and some macroscopic piece of experimental apparatus that is being used to investigate it, or you can draw it between that piece of experimental apparatus and the laboratory technician who reads its output and records it in her notebook, or you can draw it between that technician and yourself, to whom she finally submits her report, or you can draw it between your eye and your brain. But the line needs to be drawn someplace, and there need to be things on both sides of it, and so the very idea of a quantum-mechanical treatment of the universe as a whole, or of a quantum-mechanical treatment of the act of observation itself, would amount to a straightforward contradiction in terms.
Advertisement
Or something like that. The various accounts we have—as I mentioned above—are not always easy to fit together. Bohr was not particularly enthusiastic about being clear. He thought it was overrated. He is said to have taught his students that clarity and truth tend to crowd each other out, and that whereas a trivial truth is a proposition whose negation is false, a profound truth is a proposition whose negation is also true. And he seems to have been revered, for just such shenanigans, as some kind of sage.2
All of this somehow managed to harden into a rigid and powerful orthodoxy. As early as 1927, Werner Heisenberg and Max Born (both of them leading members of Bohr’s circle and future winners of the Nobel Prize) felt ready to declare that the ideas alluded to in the last several paragraphs amounted to “a closed theory, whose fundamental physical and mathematical assumptions are no longer susceptible of any modification.” From that point onward, all sorts of familiar questions about what things do and how they work were more or less universally declared to be nonsensical, and the business of inquiring any further into these matters was relentlessly discouraged. Clear and devastating arguments against the ideas of Bohr and his circle—from figures like Erwin Schrödinger, David Bohm, Hugh Everett, John Bell, and especially Albert Einstein—were met with silence, derision, inexplicable misunderstanding, or outright gibberish.3 And all of this persisted, in any number of different forms, and with undiminished zeal and intensity, for the better part of a century.
But everything that Bohr and his circle believed about these matters turns out to have been wrong. Everything that they declared to be impossible has actually been accomplished. We now have a number of promising theories of what things are “actually like” in the interiors of atoms. Each of these theories entails that the world is very different, in one way or another, from anything that we had imagined before, but what’s important for our purposes is that each of them offers us some realistic and comprehensive account of how nature objectively is, whether anybody happens to be looking at it or not. None of these theories requires that we draw any line, or make any distinction, between whatever is being measured and whatever is doing the measuring: each treats the process of measurement as a perfectly ordinary physical interaction, as something along the lines of a collision between two perfectly ordinary physical things. Each of these theories can consequently be applied to the universe as a whole. And each is compatible with everything that we currently know from experiments.
One familiar way of saying what it is that’s so puzzling about the double-slit experiment is that the electrons in them sometimes seem to act like waves (which can spread out and pass through both of the slits, and whose various parts can interfere with one another) and sometimes like particles (which remain localized and must pass through one slit or the other), depending on which particular experimental set-up we use. And many of these theories attempt to explain this behavior by picturing an electron as some sort of combination of the two.
There is a family of theories that can be traced back to Louis de Broglie and Bohm, for example, according to which electrons consist of both a particle and a wave. The electron-particles, which are always in some single determinate position in space and pass through only one or the other of the slits, are guided along their natural courses by a “pilot wave” that invariably accompanies them and that passes through both. It is the interaction between different components of these waves, after they have passed through the slits, that allows even electrons that pass through the apparatus one at a time to land on the fluorescent screen in the curious “interference” pattern of case (3), and it is the mechanical disruption of these waves by the detectors that makes it seem as if the mere act of observation can somehow make those patterns go away.
There is also a family of theories of the so-called collapse of the wave-function, which were discussed in these pages by Steven Weinberg, according to which there are only waves, but in which the waves have a tendency to occasionally and spontaneously condense into little corpuscular clumps that behave much like particles.4 The various theories in this family make slightly different predictions about the outcomes of certain experiments, all of them make slightly different predictions than the theories in the De Broglie–Bohm family, and all of them make slightly different predictions than the standard quantum-mechanical formalism of Bohr and his circle.
We don’t know yet which of these theories is ultimately going to turn out to be true—or if any of them will—and this is in part because the experiments that we would need to perform in order to find out are at present beyond our technological capacities. But there is more and more confidence, among more and more of the people who have taken the time to think carefully and seriously about these matters, that some such theory is possible and will eventually be discovered, and that the arguments that no such theory could even be imagined amounted to nothing but a particularly energetic and implacable lack of imagination.
Becker’s book is one of the first attempts we have at telling this story in a way that acknowledges how it actually turned out5—acknowledges, that is, who won these debates about the Copenhagen interpretation, who lost them, who pretended otherwise, and how they got away with it. He is sometimes a little hazy about the scientific and philosophical details of the disputes themselves. He gets mixed up, for example, about the logic of a famous argument of Einstein, Boris Podolsky, and Nathan Rosen, and about the relationship between realist and positivist and social constructivist philosophies of science. And he seems to me to give too much attention to some realistic alternatives to quantum mechanics and too little to others. But he has clearly done extensive and meticulous historical research, and his occasional missteps amount to a relatively small price to pay for the pioneering and important service he has done in finally beginning to set the record straight.
Becker is at his strongest and most persuasive when he is telling us the stories of the many lives that are wrapped up in that history. Let me briefly mention just two of those.
Einstein’s relationship to quantum mechanics is usually presented as the stuff of tragedy. He was indisputably the greatest scientist of his age. He had single-handedly overturned all earlier conceptions of space and time, and he had given us in their place, in his general theory of relativity, one of the best, most profound, and most beautiful ideas that anybody has ever had. Moreover, he himself was one of the pioneers of the new science of quantum mechanics. But he was flawed. He was proud and stubborn. He was not prepared to follow where that new science led. He was not prepared to believe that nature might refuse to accommodate itself to his intuitions.
One of the upshots of the story that Becker tells is that this is all nonsense. Einstein was out of step with his fellow physicists for the simple reason that he thought more clearly and spoke more honestly than they did. It turns out that Einstein was the first to put his finger on what was genuinely new and shocking about quantum mechanics.6 But since nobody listened to him, this important part of his achievement is only now, more than half a century after his death, and unbeknownst to his many learned biographers, finally coming into view.
The first fully developed, old-fashioned, objectively realistic alternative to the orthodox version of quantum mechanics—the one I referred to above that has both waves and particles in it—was published in the early 1950s by the American physicist David Bohm, just as he was being hounded out of the country for refusing to testify before the House Un-American Activities Committee. In 1952, when he was in exile in Brazil, a physicist named Max Dresden gave a talk about Bohm’s discoveries at the Institute for Advanced Study in Princeton, which was perhaps the most important center of theoretical physics at that time, and the reaction seems to have been vicious. According to Becker’s reconstruction, “One person called Bohm ‘a public nuisance.’ Another called him a traitor, and still another said he was a Trotskyite.”7 J. Robert Oppenheimer, the director of the institute, had earlier said of Bohm’s theory that “we consider it juvenile deviationism,” and when he was asked if he had actually read Bohm’s papers, he replied, “We don’t waste our time.” His famous advice to those assembled at Dresden’s talk was that “if we cannot disprove Bohm, then we must agree to ignore him.”
And that is what happened. Bohm had shown, decisively and for the first time, and by means of an explicit counterexample, that what Bohr and his circle had been saying was wrong. And the response—apart from one or two badly confused objections—was a cruel wall of silence. Bohm was devastated, bounced around from country to country before eventually settling at Birkbeck College in London, and seems to have decided not to mention his beautiful theory to anybody ever again.8 It languished in almost perfect obscurity until 1964, when the Irish physicist John Bell began to seriously study it and wondered why he had never heard anybody talk about it before, and did what he could, in a brilliant series of talks and papers, to make it clearer and more accessible. It wasn’t until sometime in the 1980s that a small and embattled community of physicists, mathematicians, and philosophers who had learned of the theory from Bell, began to take an active interest in what Bohm had done. His theory is now regarded as one of the two or three most important achievements in the history of our understanding of quantum mechanics.
What all these people were fighting about was the scope, ambition, and presumption of the scientific project. The story we have always been told about that dispute is that Bohr and his circle were at the vanguard of a brave and visionary intellectual upheaval, and that figures like Einstein and Schrödinger and Bohm and Bell, notwithstanding their undisputed brilliance, were somehow too timid or small-minded or mired in traditional ways of thinking to keep up.
But it turns out to have been much more complicated than that—and it might even be argued, in light of all we have learned in the meantime, that exactly the opposite was true. It might be argued that what is genuinely and permanently revolutionary about the scientific imagination is the original, unbounded, omnivorous, terrifying aspiration to reduce the entire world, and ourselves, and all our doings, to a vast concatenation of simple mechanical pushings and pullings. That’s what can never entirely be taken in. That’s what discomposes every pretense to wisdom. What Bohr and his circle were up to (in this way of looking at things) was a profoundly conservative attempt—something not altogether unlike the attempt of the Vatican three hundred years earlier, in the case of Galileo—to somehow set a limit to aspirations like that. But the business of setting such limits turned out not to be as easy as any of them had imagined.
This Issue
April 19, 2018
A Mighty Wind
The Question of Hamlet
More Equal Than Others
-
1
Bohr attached tremendous importance to the fact that we tend to describe our experimental procedures, and to report their results, in the language of the “classical” theories that quantum mechanics was supposed to replace. And that, as a matter of sociological fact, is surely true. But there is no principled argument—or none at any rate that Bohr ever presented—that seems to stand in the way of our learning to do all that, instead, in the language of quantum states. ↩
-
2
Becker quotes the American physicist John Wheeler as saying that “nothing has done more to convince me that there once existed friends of mankind with the human wisdom of Confucius and Buddha, Jesus and Pericles, Erasmus and Lincoln, than walks and talks under the beech trees of Klampenborg Forest with Niels Bohr.” ↩
-
3
Here again is Wheeler, in an astonishing letter from 1956 to a member of Bohr’s circle, pleading for tolerance on behalf of Everett, his student, who had criticized Bohr’s discussion of the act of measurement: “I have vigorously supported and expect to support in the future the current and inescapable approach to the measurement problem. To be sure, Everett may have felt some questions on this point in the past, but I do not. Moreover, I think I may say that this very fine and able and independently thinking young man has gradually come to accept the present approach to the measurement problem as correct and self consistent, despite a few traces that remain in the present thesis, draft of a past dubious attitude.” ↩
-
4
“The Trouble with Quantum Mechanics,” The New York Review, January 19, 2017. ↩
-
5
Some other notable attempts are Peter Byrne’s The Many Worlds of Hugh Everett III, Louisa Gilder’s The Age of Entanglement, and parts of Jean Bricmont’s Making Sense of Quantum Mechanics—but none of them aims at anything as comprehensive as Becker’s book does. ↩
-
6
The name of what Einstein put his finger on, by the way, was “entanglement,” which was the topic of an excellent article in these pages by Jim Holt, “Something Faster than Light? What Is It?,” November 10, 2016. ↩
-
7
Becker, to his credit, notes that the evidence for this particular reconstruction is not altogether up to his usual standards—but the story seems to have been accepted as fact by historians like Louisa Gilder. ↩
-
8
Jeffrey Bub, who is now a professor of philosophy at the University of Maryland, and who spent years at Birkbeck writing his doctoral dissertation under Bohm’s supervision, never heard of Bohm’s theory until long after his dissertation had been completed. ↩